Controlled specificity and high affinity
A tunable 100% in vitro selection process
The generation of Nanofitins® consists in identifying the best ligands amongst the vast library of variants. Libraries are screened with many parameters to tune specificity on the target(s): targeting common epitopes of different antigens (such as human/mouse cross-species binding) or forcing the competition with a ligand, particularly relevant to inhibiting a ligand/receptor interaction. This process is amenable to toxic targets, or non-immunogenic epitopes.
An extensive experience with 100+ targets
- Circulating antigens: peptides, proteins…
- Complex entities: virus-like particles, bacteria, whole cells…
- Transmembrane receptors for inhibition / modulation (GPCR, ion channels)
A common backbone with intrinsic assets
Very small and extremely stable
Nanofitins® are small, single-chain proteins (7 kDa, around 20 times smaller than a monoclonal antibody) exhibiting a higher tissue penetration potential. Nanofitins® derive from a naturally hyperstable scaffold, from which they retain most of the biochemical features such as resistance to temperature (70-80°C) and pH (10-13).
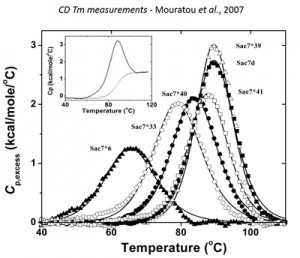
They can be stored several months at room temperature and are not sensitive to repeated freeze-thaw cycles. Nanofitins® spontaneously fold and refold, and can withstand autoclave cycles. They are not stabilized by any disulphide bridge. One major consequence is that they can be synthesised chemically. Nanofitins® have demonstrated a very high protease resistance and can thus survive in gastric fluids, which enables development of orally formulated Nanofitins® for therapeutics applications. They can also be formulated in a large spectrum of buffers and be concentrated >100 mg/mL to reach very high doses in a small volume. The combination of these features make Nanofitins® highly druggable compounds.
A promising safety profile
Toxicity
Neither cellular nor in vivo toxicity has been observed with Nanofitins®.
No mortality or clinical signs at daily repeated administration of 20 times the normal dose.
No genotoxicity or cardiotoxicity even at a high level of Nanofitin® concentration.
Biodistribution
Nanofitins® do not aggregate in kidneys; they have a rapid bladder clearance.
Immunogenicity
Nanofitins® immunogenicity has been specifically tested in several in vitro and in vivo prediction models.
No antibodies were generated against Nanofitins® after specific immunogenicity tests based upon rodent vaccination.
T-Cell assay with blood from 50 donors confirmed lack of predicted immunogenicity for Nanofitins®.
Cost-effective scale-up
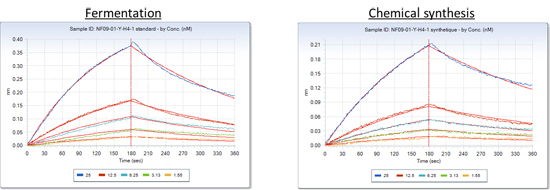
Nanofitins® can be manufactured by simple, scalable, GMP-compliant bacterial fermentation at very attractive costs. Full chemical synthesis is also possible.
Available N & C termini for simplicity of conjugation
N-/C- termini are not involved in the binding site. Thus, Nanofitins® can be :
- Easily tethered to a support for detection and capture (regioselective conjugation through insertion of one single Cysteine in the sequence),
- Conjugated to other moieties (small molecules, biologics, nanoparticles, antibodies) by genetic fusion or click chemistry
- Assembled in multimers for enhanced avidity or for multispecificity (up to 5 Nanofitins® as one multivalent compound with retained individual affinity). In every instance, neither the binding property of each single Nanofitin®, nor the function of the conjugate is affected.

